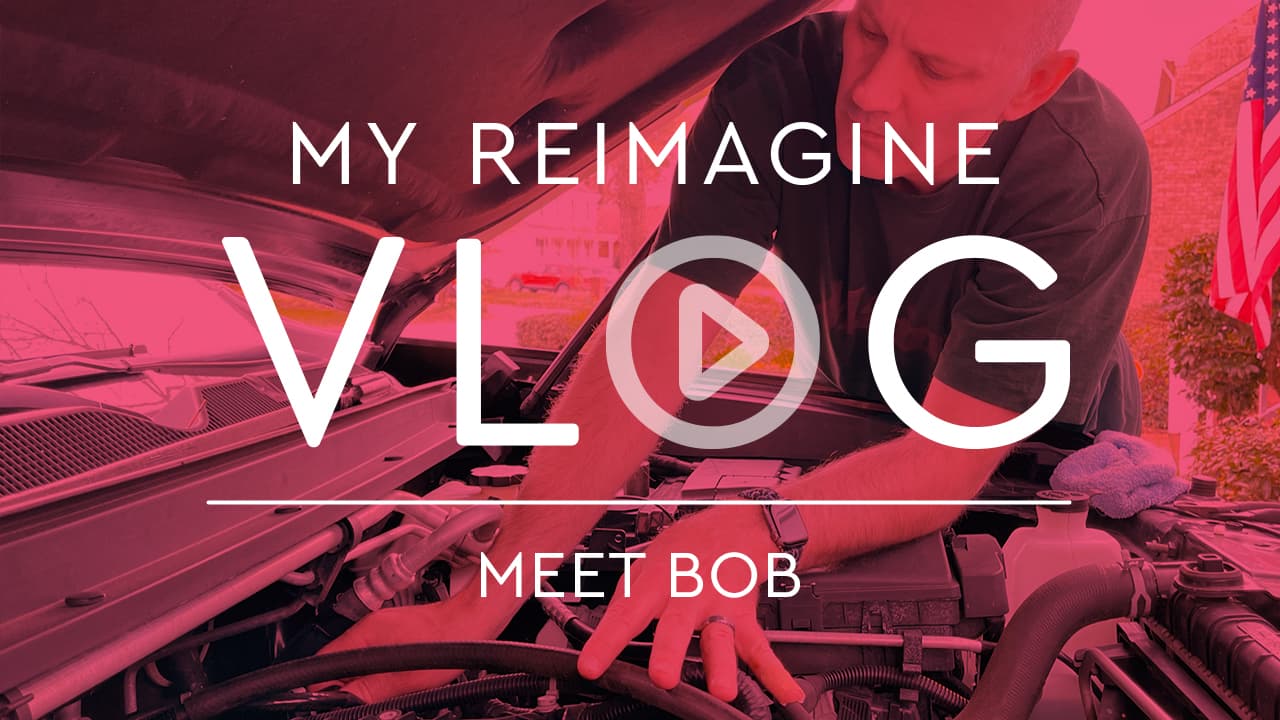3,250+ adolescents and adults have been prescribed TAKHZYRO since 2018*
Every person with hereditary angioedema (HAE) has a story to share. See how these people are choosing to reimagine their lives with TAKHZYRO. Individual experiences may vary.
*Based on third-party US specialty pharmacy data.
How did you decide TAKHZYRO was the right preventive treatment for you?
View Transcript
More Info
Learn why people trust TAKHZYRO to help reduce their HAE attacks.
DENNIS: My doctor told me about TAKHZYRO, and my wife and I did some research. At the end of the day, I trusted my medical team and what they told me. After discussing the potential risks and the possibility of fewer HAE attacks, we decided it was the right treatment to take. And it's definitely made a big difference for me. I'm glad I listened.
KELLY: Seeing the clinical data and how effective TAKHZYRO was got my attention. It told me that this is a clinically proven treatment to help prevent HAE attacks, and that gave me the confidence to start.
JACK: For me, taking TAKHZYRO once every two weeks means it's not something I need to think about often. Plus, it's subcutaneous, which means it's injected under the skin, not in the vein. That feels manageable for me.
ANDREW: The actual injection takes about a minute, and then I'm free from thinking about my next dose for a few weeks.
SORAYA: I can inject myself, and it takes about a minute. It's an important thing I can do for myself to help reduce the frequency and severity of my HAE attacks.
NARRATOR: What is TAKHZYRO? TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. Important safety information. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Please see full Prescribing Information, including information for patients, at TAKHZYRO.com. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
How did you decide TAKHZYRO was the right preventive treatment for you?
View Transcript
DENNIS: My doctor told me about TAKHZYRO, and my wife and I did some research. At the end of the day, I trusted my medical team and what they told me. After discussing the potential risks and the possibility of fewer HAE attacks, we decided it was the right treatment to take. And it's definitely made a big difference for me. I'm glad I listened.
KELLY: Seeing the clinical data and how effective TAKHZYRO was got my attention. It told me that this is a clinically proven treatment to help prevent HAE attacks, and that gave me the confidence to start.
JACK: For me, taking TAKHZYRO once every two weeks means it's not something I need to think about often. Plus, it's subcutaneous, which means it's injected under the skin, not in the vein. That feels manageable for me.
ANDREW: The actual injection takes about a minute, and then I'm free from thinking about my next dose for a few weeks.
SORAYA: I can inject myself, and it takes about a minute. It's an important thing I can do for myself to help reduce the frequency and severity of my HAE attacks.
NARRATOR: What is TAKHZYRO? TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. Important safety information. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Please see full Prescribing Information, including information for patients, at TAKHZYRO.com. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
Learn why people trust TAKHZYRO to help reduce their HAE attacks.
TAKHZYRO has been approved since 2018. What does that mean to you?
View Transcript
JACK: TAKHZYRO was studied in one of the longest preventive trials in HAE. Seeing the results gives me confidence that I'm on a treatment that not only works for me, but has been proven in clinical setting to work for others as well.
KELLY: HAE attacks are unpredictable and always will be. Knowing TAKHZYRO has been studied for as long as it was is particularly important because I'm going to have HAE for the rest of my life.
DENNIS: TAKHZYRO has been available since 2018 and doctors have prescribed it over 2500 times.
ANDREW: For me, it's not just the study results of TAKHZYRO that inspire confidence. It's also knowing that I'm on a treatment that's been prescribed to over 2500 people with HAE. It feels good to be a part of that number.
SORAYA: HAE attacks can really interfere with your life. They can happen at any time when you least expect it. The study results of TAKHZYRO tell me I'm doing something to help prevent my HAE attacks today and tomorrow.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO. The only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
Learn why these real people with HAE feel confident in their choice to start TAKHZYRO.
How has injecting TAKHZYRO been for you?
View Transcript
JACK: Injecting TAKHZYRO has not been a problem for me. For one, I only have to inject it once every two weeks. Plus, it's subcutaneous, which means it's under the skin, not in the vein. That feels manageable for me.
KELLY: Being able to switch up injection sites is a big plus for me personally, that and the fact that I can go two weeks between injections.
SORAYA: After I was trained how to do it, injecting has been manageable for me. I can do it myself, and it takes about a minute. It's an important thing I can do for myself to help reduce the frequency and severity of my HAE attacks.
DENNIS: I was having a hard time prepping the medication the way the OnePath nurse showed me. Luckily, my wife was able to do it, and I just watched her a few times. And now I am proud to say I can do it myself.
ANDREW: The actual injection takes about a minute, and then I'm free from thinking about my next dose for two weeks.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms, wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant and breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
Get some tips from people experienced with taking TAKHZYRO.
What do the 2020 US HAEA treatment guidelines mean to you?
View Transcript
JACK: The HAEA is a nonprofit advocacy and research group that helps people with HAE and it's an organization that I trust. Their website is one of the first ones I tell people to visit if they want to learn about the condition.
KELLY: The HAEA worked with experts in the medical community to create HAE treatment recommendations. These guidelines recommend that everyone with HAE talk to their doctor about long-term preventive treatment in order to find out if it's right for them.
ANDREW: My HAE attacks weren't always frequent, but they could certainly be unpredictable. I thought it was enough to just take my on-demand medication when I needed it and just push through the attack. But talking to my doctor and understanding the guidance from the HAEA about long-term prevention really helped me turn a corner in how I manage my attacks.
DENNIS: It definitely gives me confidence knowing that they recommend TAKHZYRO as a preventive option. It means TAKHZYRO is one of the preventive treatments you should talk to your doctor about.
SORAYA: The HAEA recommendation makes me think I'm taking a medication that is recommended in the HAE community and I can feel good about that.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
Learn what people taking TAKHZYRO think about their treatment being recommended by a trusted organization.
Get helpful tools for taking TAKHZYRO
What keeps you motivated to stick with TAKHZYRO?
View Transcript
More Info
See how noticing the difference TAKHZYRO makes helps keep these people motivated.
Explore what effective prevention can mean.
KELLY: I stay on treatment because I remember what my HAE was like before prevention, and I do not want to go back there.
JACK: HAE attacks are unpredictable. TAKHZYRO has made my HAE attacks less frequent. And let's face it, having fewer HAE attacks is just as important today as it was when I started treatment. I definitely don't want to go back to a life with more HAE attacks.
DENNIS: Injecting TAKHZYRO once every two weeks is certainly better than dealing with all the HAE attacks I used to have.
ANDREW: There were times before I started TAKHZYRO when all I could do was take my on-demand medication and push through the HAE attack, waiting for the medicine to work. Now that I'm having fewer attacks, I don't have to do that as much.
SORAYA: I can't say enough about the OnePath Product Support staff. They have been with me through the whole process. Whenever there was a problem accessing my TAKHZYRO, they would say, we are here to help you, and they always did. They work very hard to make sure I got my TAKHZYRO.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
What keeps you motivated to stick with TAKHZYRO?
View Transcript
KELLY: I stay on treatment because I remember what my HAE was like before prevention, and I do not want to go back there.
JACK: HAE attacks are unpredictable. TAKHZYRO has made my HAE attacks less frequent. And let's face it, having fewer HAE attacks is just as important today as it was when I started treatment. I definitely don't want to go back to a life with more HAE attacks.
DENNIS: Injecting TAKHZYRO once every two weeks is certainly better than dealing with all the HAE attacks I used to have.
ANDREW: There were times before I started TAKHZYRO when all I could do was take my on-demand medication and push through the HAE attack, waiting for the medicine to work. Now that I'm having fewer attacks, I don't have to do that as much.
SORAYA: I can't say enough about the OnePath Product Support staff. They have been with me through the whole process. Whenever there was a problem accessing my TAKHZYRO, they would say, we are here to help you, and they always did. They work very hard to make sure I got my TAKHZYRO.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
See how noticing the difference TAKHZYRO makes helps keep these people motivated.
Explore what effective prevention can mean.
What advice would you give people starting TAKHZYRO?
View Transcript
KELLY: The advice I would give someone starting TAKHZYRO is to be patient. It's easy to want a quick fix, especially when dealing with something that can be as painful and uncomfortable as HAE attacks. My doctor explained it can take about 6 doses or 10 weeks for TAKHZYRO to become constant in the body.
SORAYA: I would say that the most important thing when starting TAKHZYRO is to learn as much as you can about it. What to expect and how to take it, as well as its efficacy and side effects. Also keep talking to your doctor about any doubts or concerns you may have.
ANDREW: Even people who don't like needles should know that doing the injection takes about a minute, and it could help reduce your HAE attacks. Also, the HAEA and OnePath Patient Support Managers are excellent resources that should be used.
DENNIS: I tell people to imagine what their lives could be like with fewer HAE attacks. That's what taking TAKHZYRO did for me.
JACK: My advice would be to stick with it. Remember what your HAE attacks were like before taking a preventive treatment. And take the time to recognize what's different about your HAE attacks since starting treatment.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms, wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE you take as a subcutaneous injection, just once every two weeks.
Get tips for starting on TAKHZYRO and making the most of support, from real people who've been where you are.
This is my TAKHZYRO Experience: Imagine Yours
View Transcript
JACK: When I was first prescribed TAKHZYRO, my insurance company denied it. But I worked with my doctor and OnePath Product Support stepped in to help too. Thanks to them, I never missed a dose.
DENNIS: When I started taking TAKHZYRO, I had some soreness around my injection area. Told my doctor about it. He said, "Give it some time." And after a few injections, I stopped having that soreness. The important thing is I'm having fewer HAE attacks than before.
SORAYA: During my first month taking TAKHZYRO, I had an HAE attack. I talked to my doctor, and he said, "This is normal. The medicine builds in your body as you take it." And after a few months, I started to feel the difference in my HAE attacks. They were less frequent and less severe, and now I am so happy I stuck with it.
KELLY: My doctor explained that TAKHZYRO takes about 10 weeks to build up and become constant in your body. Once it does, it's called steady state. I was eager to get a steady state.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms, wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1 (800) FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
Hear real people share their experience starting TAKHZYRO.
Need help remembering when to take TAKHZYRO?
Why is it important to set treatment goals?
View Transcript
ANDREW: Setting goals for your preventive treatment helps you to see your progress.
DENNIS: It helps to have realistic goals for your treatment. Some people may want zero HAE attacks, but for me, just having fewer and less severe HAE attacks was my goal. And when I achieved that goal, I knew TAKHZYRO was the right choice for me.
SORAYA: I track things like how many HAE attacks I'm having, where they are, and how severe they are. I've gone two months without an HAE attack. My goal is to continue reducing the frequency of those attacks.
JACK: I also track how often I use my on-demand treatment because if I'm using it less, that's a sign for me that TAKHZYRO is doing what it's supposed to do, helping prevent HAE attacks.
KELLY: I reviewed my HAE management goals with my doctor at my six-month appointment after starting TAKHZYRO just to make sure I was tracking where I wanted to be.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema, HAE, in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms, wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1 (800) FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed.
NARRATOR: Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
There are a few great ways to track your progress. Listen to how and why others have set goals for HAE management.
Find the inspiration you need to stay on track.
What advice do you have for other caregivers?
View Transcript
BOBBI: For other caregivers dealing with HAE, I think the biggest thing you could do is just be supportive and try to understand even if you don't truly understand what that person is going through. But just be there, and love them through it.
JASON: My advice to a new caregiver is to ask a lot of questions, get a lot of answers, and don't be scared to ask questions. There's a lot of people out there that can help, have answers, that they have gone through in their life. It may help them out.
PAM: I would tell caregivers first of all, if you don't have HAE, is to find out about it, to-to know what's going on, and what things need to be done because sometimes those attacks can be an emergency or a life-threatening situation. So, you need to be prepared about what needs to happen for the person who is having the attack. And then I think it's to be sensitive to what's going on with them, to find out or look for clues as to when they might be having attacks coming on, and they may not even recognize it themselves.
JUAN: What I would like say to other caregivers is don't lose hope. There is support out there. There is help out there, and there's a community of people that have gone through the same exact situation that you are in, and there are people that you can talk to.
NARRATOR: What is TAKHZYRO? TAKHZYRO® (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema (HAE) in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age.
NARRATOR: TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives. The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist.
NARRATOR: You may report side effects to FDA at 1-800-FDA-1088. TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every two weeks.
Educate yourself, be prepared, remember there’s a community of support out there—hear more thoughts and advice from fellow caregivers.
Wondering what to expect when a loved one starts TAKHZYRO?
Jack's Vlog
View Transcript
More Info
Jack knows how unpredictable HAE can be, as he and 13 members of his family have been diagnosed with the disease. When deciding to take TAKHZYRO, Jack made sure to discuss his goals and expectations for his HAE management plan with his doctor.
Helpful tips:
- Pay attention and be aware of what you're missing out on due to HAE attacks by keeping track in a journal
- Looking for a community like Jack? Check out HAEA.org and attend advocacy meetings or events
Get ideas to have a productive conversation with your doctor
JACK: Hi, my name is Jack, and this is My Reimagine Vlog.
JACK: Hello. My name is Jack, and I've been taking TAKHZYRO for more than 2 years. Hereditary angioedema, or HAE, has been a large part of my family's life. Thirteen members of my immediate and extended family have been diagnosed with HAE. Because growing up I knew and cared about so many people with this rare disease, it inspired me to want to take care of others. I was actually diagnosed with HAE at 13, but I didn't have any severe symptoms until much later in life. Some of my family members were so much worse off than I was and I found myself putting to the side that I had HAE as well. I tried to help out as much as any kid could – I’d go to the hospital with my mom, and I'd sit with her while the doctors tried to find a way to manage her pain. From that point on, I made it one of my primary goals in life to pursue a medical career in which I could help other patients. But achieving that was a challenge, as my goals and expectations changed and evolved along the way.
JACK: Fortunately, or unfortunately, my HAE didn't become apparent until I was in nursing school. I started to get swells, and it seemed that stress started to trigger my HAE. The attacks came every few months at first, but then they progressed to every other month. Eventually, I was experiencing attacks every month. After graduation, I got a job as a pediatric oncology nurse, but at this point in my life, my attacks were becoming more frequent and more severe. I wasn't sure how I was going to keep my attacks under control, and it was hard to stay focused at my job. It got to the point where I missed so much work that I was asked to step down from my position.
JACK: I used this time to get really involved with the HAE community. I did some advocacy work. I was mentoring young children with HAE. The more I was involved in the community, the more I learned about HAE and about the treatment options available to us patients. I wasn't interested in trying any preventive medications at the time, but as each year passed, I realized my attacks were becoming more frequent and more severe, and it became clear that I needed to take a closer look at preventive therapies that might be able to help. I wanted a treatment that could reduce the amount of swells I was having. I think fate intervened when I learned about TAKHZYRO during one of my regular doctor's visits. A nurse told us that TAKHZYRO, a once-every-2-weeks subcutaneous injection used to prevent HAE attacks, had been approved.
JACK: Right then and there, my doctor and I talked about it. We also discussed what to expect and set goals to manage my HAE. Reducing my attack frequency was the primary goal–from having swells every week to less frequently. We also discussed the potential risks such as serious side effects, including allergic reactions, as well as more common side effects, including injection site reactions, upper respiratory infections, and headaches. As far as allergic reactions, my doctor told me to call or get emergency help right away if I experienced any of the following: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, or hives.
JACK: Since starting TAKHZYRO, the frequency and severity of my attacks have decreased. I was accustomed to multiple attacks a week. After starting TAKHZYRO, I've gone a month or two without an attack. I even had a span as long as 6 months when I had zero attacks. And if I do experience an attack, it's usually less severe than before. This is just my experience with TAKHZYRO, and it may not be the same for everyone. 2020 was one of the most stressful years of my life, and like I said earlier, stress is one of my triggers. I built a new house, so my wife and I had to deal with the stress of moving. On top of that, being a nurse during the COVID-19 pandemic has not been easy, to say the least. With all of this happening, I'm glad I had a management plan for my HAE.
JACK: These days, my wife and I are discussing a new goal: starting a family. We're ready to see how we can fill our new place with more joy. And I've been spending my free time exercising. I feel like it's a great way for me to get back into shape, and it helps that we encourage each other to stay fit and follow our workout goals.
JACK: When I was first managing my HAE, all I wanted was to simply reduce the amount of swells from weekly to maybe once a month. After that, the goal was to not go to the hospital as often. Setting goals and expectations is very important. I did it for myself when it came to my career, and I did it for my HAE management plan. I encourage you to talk to your doctor about your individual HAE goals and expectations. This journey has taken me through many ups and downs, and things have evolved along the way. Now I feel my HAE is better managed. I know one goal I can continue to work closely on with my doctor: reimagining life with HAE.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema (HAE) in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088.
NARRATOR: TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every 2 weeks.
Jack's Vlog
View Transcript
JACK: Hi, my name is Jack, and this is My Reimagine Vlog.
JACK: Hello. My name is Jack, and I've been taking TAKHZYRO for more than 2 years. Hereditary angioedema, or HAE, has been a large part of my family's life. Thirteen members of my immediate and extended family have been diagnosed with HAE. Because growing up I knew and cared about so many people with this rare disease, it inspired me to want to take care of others. I was actually diagnosed with HAE at 13, but I didn't have any severe symptoms until much later in life. Some of my family members were so much worse off than I was and I found myself putting to the side that I had HAE as well. I tried to help out as much as any kid could – I’d go to the hospital with my mom, and I'd sit with her while the doctors tried to find a way to manage her pain. From that point on, I made it one of my primary goals in life to pursue a medical career in which I could help other patients. But achieving that was a challenge, as my goals and expectations changed and evolved along the way.
JACK: Fortunately, or unfortunately, my HAE didn't become apparent until I was in nursing school. I started to get swells, and it seemed that stress started to trigger my HAE. The attacks came every few months at first, but then they progressed to every other month. Eventually, I was experiencing attacks every month. After graduation, I got a job as a pediatric oncology nurse, but at this point in my life, my attacks were becoming more frequent and more severe. I wasn't sure how I was going to keep my attacks under control, and it was hard to stay focused at my job. It got to the point where I missed so much work that I was asked to step down from my position.
JACK: I used this time to get really involved with the HAE community. I did some advocacy work. I was mentoring young children with HAE. The more I was involved in the community, the more I learned about HAE and about the treatment options available to us patients. I wasn't interested in trying any preventive medications at the time, but as each year passed, I realized my attacks were becoming more frequent and more severe, and it became clear that I needed to take a closer look at preventive therapies that might be able to help. I wanted a treatment that could reduce the amount of swells I was having. I think fate intervened when I learned about TAKHZYRO during one of my regular doctor's visits. A nurse told us that TAKHZYRO, a once-every-2-weeks subcutaneous injection used to prevent HAE attacks, had been approved.
JACK: Right then and there, my doctor and I talked about it. We also discussed what to expect and set goals to manage my HAE. Reducing my attack frequency was the primary goal–from having swells every week to less frequently. We also discussed the potential risks such as serious side effects, including allergic reactions, as well as more common side effects, including injection site reactions, upper respiratory infections, and headaches. As far as allergic reactions, my doctor told me to call or get emergency help right away if I experienced any of the following: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, or hives.
JACK: Since starting TAKHZYRO, the frequency and severity of my attacks have decreased. I was accustomed to multiple attacks a week. After starting TAKHZYRO, I've gone a month or two without an attack. I even had a span as long as 6 months when I had zero attacks. And if I do experience an attack, it's usually less severe than before. This is just my experience with TAKHZYRO, and it may not be the same for everyone. 2020 was one of the most stressful years of my life, and like I said earlier, stress is one of my triggers. I built a new house, so my wife and I had to deal with the stress of moving. On top of that, being a nurse during the COVID-19 pandemic has not been easy, to say the least. With all of this happening, I'm glad I had a management plan for my HAE.
JACK: These days, my wife and I are discussing a new goal: starting a family. We're ready to see how we can fill our new place with more joy. And I've been spending my free time exercising. I feel like it's a great way for me to get back into shape, and it helps that we encourage each other to stay fit and follow our workout goals.
JACK: When I was first managing my HAE, all I wanted was to simply reduce the amount of swells from weekly to maybe once a month. After that, the goal was to not go to the hospital as often. Setting goals and expectations is very important. I did it for myself when it came to my career, and I did it for my HAE management plan. I encourage you to talk to your doctor about your individual HAE goals and expectations. This journey has taken me through many ups and downs, and things have evolved along the way. Now I feel my HAE is better managed. I know one goal I can continue to work closely on with my doctor: reimagining life with HAE.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema (HAE) in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088.
NARRATOR: TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every 2 weeks.
Jack knows how unpredictable HAE can be, as he and 13 members of his family have been diagnosed with the disease. When deciding to take TAKHZYRO, Jack made sure to discuss his goals and expectations for his HAE management plan with his doctor.
Helpful tips:
- Pay attention and be aware of what you're missing out on due to HAE attacks by keeping track in a journal
- Looking for a community like Jack? Check out HAEA.org and attend advocacy meetings or events
Get ideas to have a productive conversation with your doctor
Bob's Vlog
View Transcript
BOB: Hi, my name is Bob. And this is My Reimagine Vlog.
BOB: Hello. My name is Bob, and I've been taking TAKHZYRO for more than 2 years. For me, I've always been about pushing myself to get the most out of life. When I was 19, I joined the Marines, and being in the Marines was all about pushing yourself to perform at your peak. However, there came a point where I couldn't push any further. I was suffering from abdominal attacks, and I didn't know why.
BOB: Nine years into my career as a Marine, I found out that my sister was diagnosed with hereditary angioedema, or HAE. Knowing that, I also got tested and found out that I have HAE as well. The good news was that I finally found out that there was a reason why my body was experiencing all these attacks. But the bad news? After my diagnosis, the career that I was building was suddenly over.
BOB: Where could I push myself further? I don't think I could have pushed myself to become who I am now if I was still in the Marines. It was a blessing in disguise. Since I was no longer enlisted, I had the chance to go on a blind date. And now, 25 years later, that blind date has resulted in having a beautiful wife and 2 children.
BOB: I tried to manage my HAE attacks throughout the years, but my attacks kept coming, sometimes without rhyme or reason. Whenever I was stressed, I would get attacks. Whenever I was just stressed about stress, I would get attacks. HAE was getting the best of me, and I wasn't satisfied with my HAE management plan. Sometimes, my abdominal attacks were crippling and required visits to the ER. I remember one year when I had to go to the ER 6 times. Every time I went in, I would have to re-explain my condition to whoever was on duty while I was in mid-attack. I was being labeled as someone coming in for pain medication.
BOB: Eventually, in September of 2018, my doctor told me about TAKHZYRO. It's a subcutaneous injection that you take just once every 2 weeks to prevent HAE attacks. He was very clear from the beginning about the clinical trial results, and he said that there was a real possibility that TAKHZYRO could help decrease the frequency and severity of my attacks. We also discussed the potential risks, such as serious side effects, including allergic reactions as well as more common side effects, including injection site reactions, upper respiratory infections, and headaches. My doctor told me to call or get emergency help right away if I experienced any of the following symptoms from an allergic reaction, including wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, or hives.
BOB: What was interesting for me is what my doctor told me about how TAKHZYRO performed in clinical studies. Some patients taking TAKHZYRO 300 milligrams once every 2 weeks experienced on average an 87% reduction in attacks at 6.5 months versus those who took the placebo. Not only that, but TAKHZYRO helped patients reduce moderate or severe attacks, as well as attacks that required acute treatments.
BOB: I've experienced this in real life, though I still keep my acute treatment handy. Some patients in the clinical study also reached zero attacks for periods of time. Prior to using a preventive treatment, I was experiencing 2 to 3 attacks per month. Now with TAKHZYRO, I've gone up to 12 months without an HAE attack. These are my experiences, and I realize every patient's experience is different. So talk to your doctor about TAKHZYRO and if it may be appropriate for you.
BOB: Lately, I've enjoyed playing golf with my wife and daughter. We're practicing our putting wherever we can, just enjoying ourselves, slowly improving our skills at the game. One thing I've always wanted to do is restore an old car. I finally had an opportunity to do that this past year. My friend and I took a vintage 1970s car and turned something old new again, from how it used to run to how it runs now. We've reimagined what was possible for that car, giving it a second opportunity.
BOB: My experience with TAKHZYRO has given me the possibility of fewer attacks. If you're interested in learning more about reimagining your life with HAE, talk to your healthcare provider to see if TAKHZYRO is right for you.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema (HAE) in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088.
NARRATOR: TAKHZYRO has not been studied in pregnant or breastfeeding women. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed. Please see full Prescribing Information, including information for patients, at TAKHZYRO.com. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every 2 weeks.
Bob has always pushed himself to get the most out of life, but his HAE was getting the best of him. When his doctor told him TAKHZYRO could reduce the frequency of his attacks, Bob decided to give it a try – and more than two years later, Bob is continuing to reimagine his life with HAE.
Helpful tips:
- Notice stress being a trigger like Bob? Try journaling before and after an attack to help notice what may be triggering you
- Bob found himself going to the ER with no plan. Fill out the HAE attack plan so you have one if something similar happens
Explore what effective prevention can mean
Kelly's Vlog
View Transcript
KELLY: Hey, my name is Kelly, and this is My Reimagine Vlog.
KELLY: Hello, my name is Kelly. I live in a small town in North Carolina, and I work as a branch manager at a bank. I've been taking TAKHZYRO for more than 2 years. The first time I went hiking was when I was in middle school–there was a trail with a bunch of stairs that led to the top of a mountain. I remember reaching the top and the amazing sight and seeing everything and thinking wow, I made it. When I woke up the next day after the hike, I had swollen legs and swollen feet. The thing is, I was diagnosed with hereditary angioedema, or HAE, when I was 7, but I never let my condition stop me from doing what I wanted to do. I especially didn't let it stop me from my love of hiking, and I kept at it through the years. Growing up with HAE was always an uphill challenge for me. Kids were harsh, and I was teased a lot.
KELLY: I remember going home one day and crying and not knowing what to do. Well, when I had swells at school, if there were things that I could hide, whether it was, you know, my feet or my legs, I just didn't talk about it or draw attention to it. But it was when I had swells at school that I couldn't hide, whether it would be my hands or my face. Luckily, I have an older brother who has always been there for me. He brought a crown over that night, a huge one, and told me to put it on my head. He told me to look down, and of course when I did, the crown slid off my head. He asked me to pick it up off the ground and wear it again—this time holding my head high. My brother said: "This crown represents your self-worth and your confidence. You should be able to walk into every room with your head held high, because you deserve respect and love. I want you to own your disease, your HAE."
KELLY: Those words have never left me, and I began to accept that fact. I had HAE, I couldn't change that fact, but I didn't have to be ashamed of it. It was still an uphill journey through the years, trying to find the right treatment for me, and I was still having attacks 1 to 3 times a week. Sometimes they were so severe that I had to go to the hospital. I knew that I wanted to find a preventive treatment because my HAE was getting in the way of how I wanted to live, so I did some research, and I found out about TAKHZYRO. I actually brought it up with my doctor.
KELLY: I was interested in TAKHZYRO because it was a once-every-2-weeks subcutaneous injection used to prevent HAE attacks. Of course, before making the decision to start TAKHZYRO, my doctor and I discussed several potential risks such as serious side effects, including allergic reactions, as well as more common risks, including injection site reactions, upper respiratory infections, and headaches. My doctor was very specific with me about the symptoms of an allergic reaction and told me to call or get emergency help right away if I ever experienced any of the following: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, or hives. We also discussed how TAKHZYRO could help reduce the frequency and the severity of my attacks. Sometimes on my hikes, before I reach the top, there are rest areas or viewpoints. When I first started TAKHZYRO, my doctor told me it wouldn't instantly work.
KELLY: There's this thing called "steady-state" that your doctor might talk to you about. What that means is that it takes time for TAKHZYRO to get to a constant level in the body. You have to have patience to wait for TAKHZYRO to reach that “steady-state”. About 10 weeks is around the time that my doctor said TAKHZYRO would take to build up fully in my body. Steady, state. Know that the rest areas or viewpoints are just stops along the way, and it's important to keep moving forward. Since starting TAKHZYRO, I've experienced less frequent attacks. I still experience attacks on treatment with TAKHZYRO, but the severity of my attacks decreased. I've also experienced several attack-free months since starting TAKHZYRO. Every patient's experience is different, so it's important to work with your doctor to see if TAKHZYRO would be a good option for you. I think about the first time I went hiking and where I am today. I have a whole new perspective now through my journey with HAE and TAKHZYRO. I am who I am because of the challenges I faced, and I am where I am because I kept my head up.
KELLY: Sometimes the longest hikes are the most rewarding, and TAKHZYRO has helped me reimagine my life with HAE.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema (HAE) in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088.
NARRATOR: TAKHZYRO has not been studied in pregnant or breastfeeding women. Please see full Prescribing Information, including information for patients, at TAKHZYRO.com. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every 2 weeks.
Kelly wanted to find a preventive treatment because of her frequent HAE attacks. When she decided to start TAKHZYRO, her doctor told her it wouldn't instantly work, so she had patience and continued to move forward to reimagine her life with HAE.
Helpful tips:
- Don't forget to ask your doctor to talk to you about steady state
- Think about the hobbies you'd like to focus on after you've been on TAKHZYRO for a while
Find tips for staying on track with TAKHZYRO
Jenny's Vlog
View Transcript
JENNY: Hi, my name is Jenny and this is My Reimagine Vlog. David, are you ready?
DAVID: Yeah.
JENNY: For some spinning?
DAVID: Oh, yeah.
JENNY: Hello, my name is Jenny, and I've been taking TAKHZYRO for more than 2 years. However, I don't think I would have made it this far if not for my caregiver, partner, and husband Juan. It's certainly been a long journey with hereditary angioedema, or HAE, but I've had him with me for a large part of it. He’s seen me when my attacks are so bad that I have to stay in bed and move as little as possible. When I just have to sleep it off and I'm out of commission for days.
JUAN: How did this relationship start? Jenny and I met in Spain where we were college students. Jenny was studying abroad, and she asked me out on our first date.
JENNY: True story. Juan is from Spain and I asked him out for not like a date, date. It was a, "Hey, I'm here studying Spanish. You clearly speak English, so let's do an Intercambio." So it was a total language-geek first date.
JUAN: All I remember was, did a cute girl just start asking me out? "Yep, I'll be there." We had lived a long-distance relationship for so many years before we got married. Got married in Spain, got a teeny tiny little apartment, maybe like half an hour away from the Mediterranean, dog, life, you know. But we did want to start a family. So, you know, did a balance, what should we do? So we decided, packed it all up. And here we are a few years later. I mean, we exchanged a little house right away from the Mediterranean to living in New England winters. But I can't say that's too bad of trade.
JENNY: Oh, so sweet.
JENNY: Despite being on a preventive medication already, when I heard about Takeda's preventive treatment TAKHZYRO, a once-every-2-weeks subcutaneous injection, I used this as an opportunity to reach out to my doctor and discuss my HAE management plan.
JENNY: We both took a hard look at the clinical trial data, and we thought it might offer me a chance to experience less severe attacks compared to before being on preventive medication. We discussed the potential risks associated with TAKHZYRO. And my doctor went through the serious side effects, including allergic reactions, as well as more common side effects, including injection site reactions, upper respiratory infections, and headaches. We discussed the symptoms of an allergic reaction. And my doctor told me to call or get emergency help right away if I experienced any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, or hives.
JENNY: After discussing everything at length, I felt comfortable moving forward with TAKHZYRO as my preventive medication. When I started TAKHZYRO, it wasn't exactly smooth sailing. There was a transition period for a few months when I was still getting frequent swells, and I was questioning if TAKHZYRO was right for me. I had to stay home during that time, and Juan would take off work to take care of the boys. Whenever I got an abdominal attack, I would be physically drained. I felt like a battery that was at 0%. But I pushed through with rest, self-care, and by asking for help. It's important to have help. You should not be afraid to ask for it when you need it.
JUAN: And I reminded Jenny that she made this choice carefully with her doctor and I and to stay strong. One consideration that was important to Jenny when she started TAKHZYRO is that it is taken every 2 weeks and that the injection only takes up a small part of her day. I would encourage anyone who is beginning treatment with TAKHZYRO to talk to their doctor about what to expect when they first start and how long it will take for TAKHZYRO to get to a constant level in the body. Seeing Jenny in pain was not easy. And I knew I had to stay strong for our family, but I'm really glad she stuck with it.
JENNY: I continued to take TAKHZYRO, and now, more than 2 years later, my attack frequency and severity have decreased. I still get abdominal attacks once in a while, but I have my acute treatment with me at all times, so I'm prepared. But these are my experiences, and not everyone's experiences with TAKHZYRO will be the same.
JENNY: Knowing that HAE is a genetic condition, we got our sons tested as early as possible. David, our oldest, tested negative, but Sebastian tested positive for HAE in September 2019. Thoughts of guilt flooded my mind after I received that dreadful call. I cried, and I cried a lot. I had to pick up the boys from daycare, and on the drive there, I managed to call Juan while he was at work. The words barely made their way out of my mouth, but they eventually did: "Sebastian has HAE." A long pause followed. I could hear Juan start to cry. I didn't know exactly what he was thinking, but my mind reverted back to every painful attack, every visit to the hospital, and every tear shed because of my HAE. I felt guilty for giving my son something that would cause him pain. No mother ever wants to cause their child pain.
JENNY: Fortunately, there are several resources and treatments available to patients with HAE. I was able to find a treatment that helps prevent my own HAE attacks. When the time comes, Sebastian will have appropriate options available as well.
JUAN: It's been great to see Jenny live her life. She's not just been an incredible mother to David and Sebastian, but she's also been teacher Jenny to our nieces during this pandemic year. For 3 days during the week, she holds classes for them, and around the holidays, they made their own gingerbread houses, and Jenny judged them on their creativity. But she also made sure to take points off if the kids ate the building supplies.
JENNY: Don't eat the gingerbread.
JUAN: Or the marshmallows.
JENNY: I mentioned at the beginning that I don't think I would have made it this far if not for Juan being in my life. His role as a husband and caregiver, along with my management plan that includes TAKHZYRO, has changed my journey with HAE. Just asking for help and allowing him to care for me was a big step in my life. And I know that he will always be there for me and our boys. Juan goes beyond just giving care; Juan gives me hope and the possibility to live life beyond my HAE.
NARRATOR: TAKHZYRO (lanadelumab) is a prescription medicine used to prevent attacks of hereditary angioedema (HAE) in people 2 years of age and older. It is not known if TAKHZYRO is safe and effective in children under 2 years of age. TAKHZYRO may cause serious side effects, including allergic reactions. Call your healthcare provider or get emergency help right away if you have any of the following symptoms: wheezing, difficulty breathing, chest tightness, fast heartbeat, faintness, rash, hives.
NARRATOR: The most common side effects seen with TAKHZYRO were injection site reactions (pain, redness, and bruising), upper respiratory infection, headache, rash, dizziness, diarrhea, and muscle aches. These are not all the possible side effects of TAKHZYRO. For more information, ask your healthcare provider or pharmacist. You may report side effects to FDA at 1-800-FDA-1088.
NARRATOR: TAKHZYRO has not been studied in pregnant or breastfeeding women. Please see full Prescribing Information, including information for patients, at TAKHZYRO.com. Talk to your healthcare provider about the risk of taking TAKHZYRO if you are pregnant, plan to be pregnant, are breastfeeding, or plan to breastfeed. Talk to your healthcare provider about TAKHZYRO, the only preventive HAE treatment you take as a subcutaneous injection just once every 2 weeks.
When Jenny started TAKHZYRO, it wasn’t smooth sailing: she was still getting swells and began to question if it was the right treatment for her. With the support of Juan, her husband and caregiver, she stuck with it and these days she has the possibility to live with fewer and less severe HAE attacks.
Helpful tips:
- Ask a trusted friend or loved one to listen and help you with your HAE
- Does HAE run in your family? Check out this TAKHZYRO for Children Webinar video.
Help a loved one stay on track with TAKHZYRO